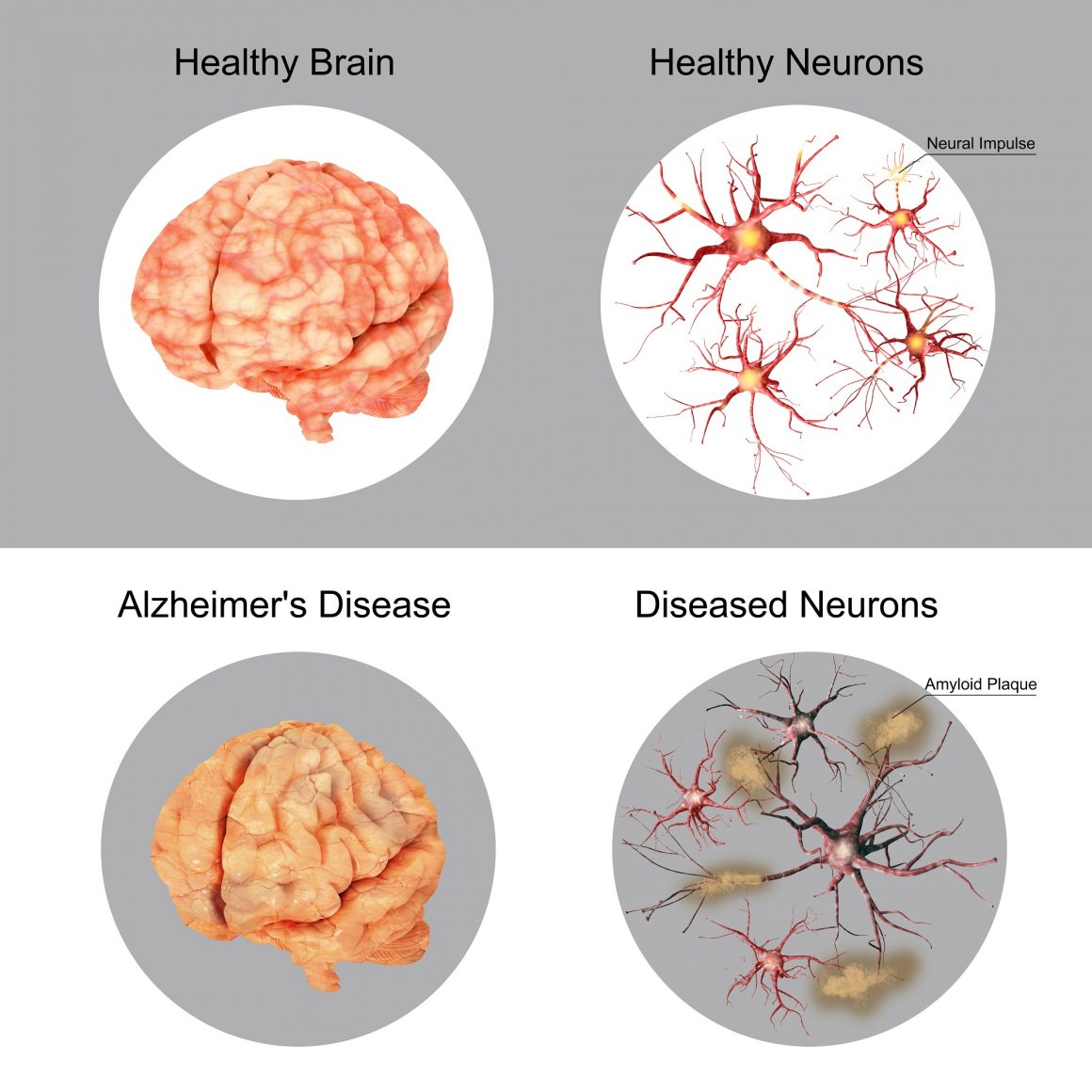
Finding a cure for Alzheimer’s Disease remains a difficult challenge for medical researchers, but as they’ve uncovered characteristics and behaviors of the disease, new treatments are being unveiled that show the potential to slow or even reverse its effects.
Some of those treatments may not be the traditional pathways toward a cure you might expect, such as rehabilitation or pharmaceutical approaches. The latest potential treatment comes in the form of a technological solution, a wearable cap from Arizona based NeuroEM Therapeutics that works to reverse cognitive impairment caused by Alzheimer’s.
Working in conjunction with the University of South Florida’s Byrd Alzheimer’s Institute, the company conducted a clinical trial to assess the safety and efficacy of a head device called MemorEM that uses a process called transcranial electromagnetic treatment (TEMT), which sends electromagnetic signals through a frequency into the brain. The result in preclinical trials has been a reversal of memory loss in animal models.
“This pioneering study suggests that TEMT may be an entirely new therapeutic intervention against Alzheimer’s disease,” said Dr. Gary Arendash, CEO of NeuroEM Therapeutics in a statement following the study’s release. “Our bioengineering technology may be succeeding where drug therapy against this devastating disease has thus far failed. TEMT appears to be affecting the Alzheimer’s disease process through several actions directly inside neurons (brain cells), which is where we believe the disease process needs to be stopped and hopefully reversed.”
How Does It Work?
The study, published in the Journal of Alzheimer’s Disease, examined how these signals could affect small collections of toxic proteins that lead to Alzheimer’s, such as beta amyloid and Tau proteins.
To do it, researchers recruited eight participants suffering from mild to moderate Alzheimer’s disease and enlisted the help of their caretakers to help participants use the MemorEM device in a home care setting. Participants received the treatment twice a day for two months, with each session lasting for one hour.
The cap itself is portable, allowing participants freedom of movement in their homes and making it easy for caregivers to ensure it is used properly.
At the conclusion of the trial, no participants had experienced any side effects. Brain scans conducted by the researchers showed that the eight individuals had not developed tumors or incurred any brain damage as a result of having used the device. This verified its safety, but what about the efficacy?
What Results Did TEMT Yield?
To measure whether or not the treatment had worked, investigators used The Alzheimer’s Disease Assessment Scale-Cognitive Subscale Trusted Source test (ADAS-cog). This is the most commonly recognized method for cognitive function assessments.
What they discovered was that seven of the eight participants increased their ADAS-cog scale scores by over 4 points after just those two months. Researchers estimate that is equivalent to the participants’ cognitive function being “rejuvenated” by a year.
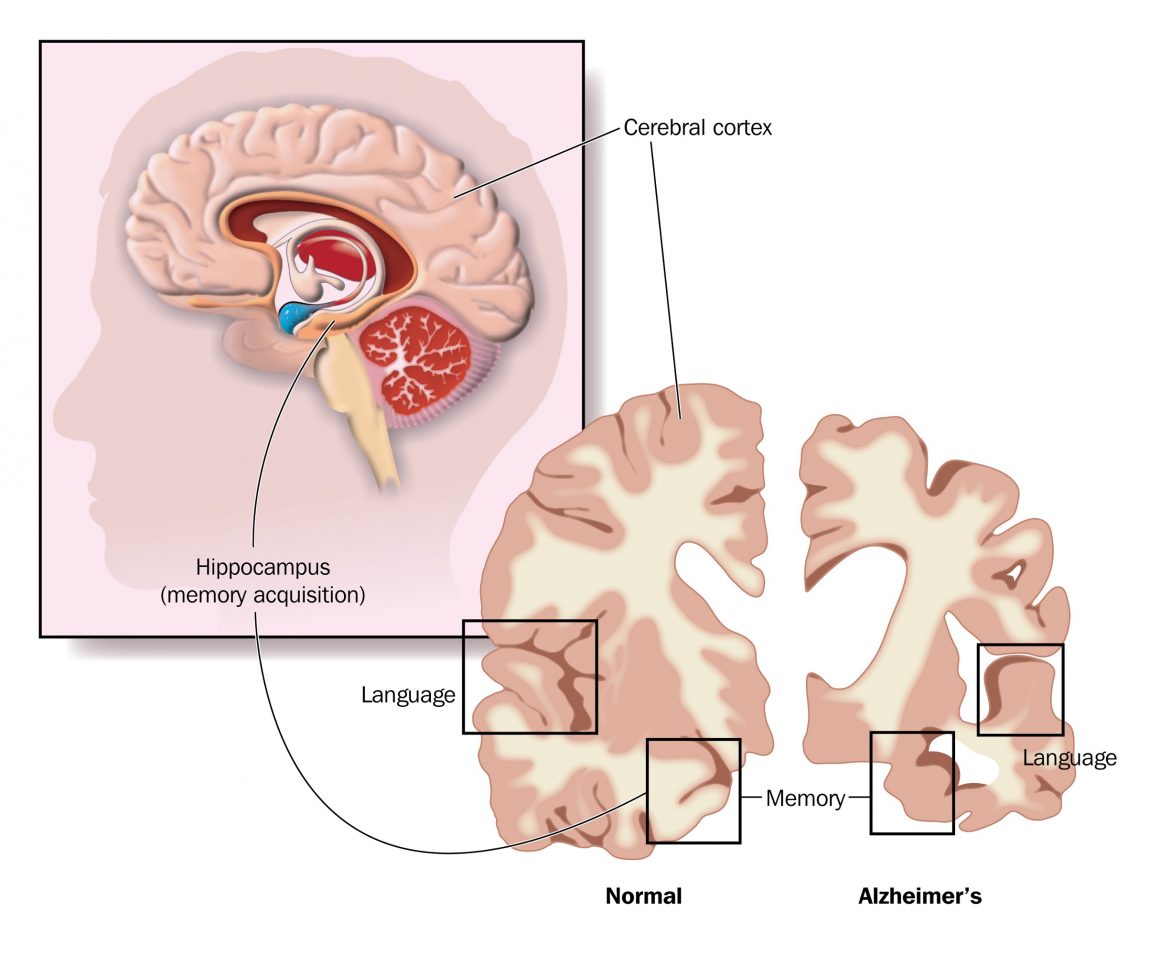
Further evaluation shows that the reason participants test scores improved was they had experienced an improvement in communication between brain cells in the cingulate cortex, an area of the brain located within the cerebral cortex.
Participants saw a 50% decrease in memory lapses, while TEMT also had a positive effect on Alzheimer’s markers in blood and the cerebrospinal fluid (CSF) around the brain.
What’s Next?
Researchers revealed that at the conclusion of the study none of the participants wanted to return the MemorEM, a good sign that patients feel like the device is having a positive effect. But this is purely anecdotal evidence.
Obviously, a study involving eight people is not all that is needed to prove the value of MemorEM, though it is a promising start. Plans are in place for a larger trial to take place involving 150 patients coping with Alzheimer’s by the end of 2019. Should that yield similar results, you can expect to see NeuroEM Therapeutics push to get the device approved by the Food and Drug Administration as a treatment for Alzheimer’s.
