New technology developed by the Israeli company BrainQ may help reduce disability in stroke victims. A hat-like device developed by the company uses artificial intelligence-based electromagnetic therapy to treat damaged areas of the brain. Early studies already show remarkable results.
It’s a breakthrough that could change how clinicians treat those who have suffered a stroke. The BrainQ device sends electromagnetic waves into damaged areas of the brain. These artificial brain waves mimic healthy neural networks. The goal is to rebalance the brain and restore normal brain function.
The innovative technology could especially have an impact in parts of the world where strokes are common, including the stroke belt in the United States.
What Happens to the Brain After a Stroke?
About 795,000 people in the United States have a stroke every year, while 15 million occur worldwide. About 87% of all strokes in the U.S. are ischemic strokes, which involves partial or complete blockage of blood flow to the brain. Brain cells begin to die within minutes.
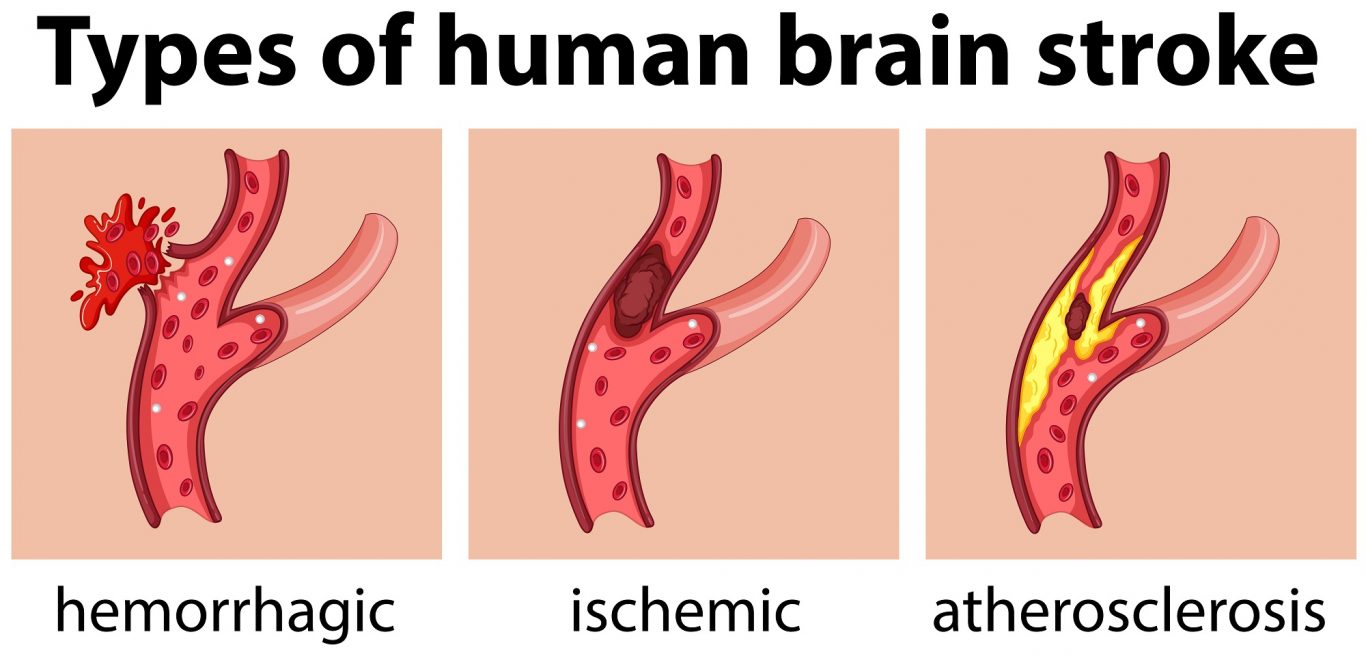
The impact of a stroke on the brain depends on where the stroke occurred. For example, according to the American Stroke Association, a stroke in the left brain will cause paralysis on the right side of the body, speech/language problems, slow and cautious behavior and memory problems.
A stroke on the right side of the brain will also cause memory issues, as well as paralysis on the left side of the body, vision problems and a quick, inquisitive behavior. A stroke in the back of the brain will likely impact vision, while a stroke in the brain stem can cause paralysis on both sides of the body.
A Small Study Shows “Striking Result”
BrainQ hopes its new device will help stroke victims recover from the impact of a severe stroke. Yotam Drechsler, the company’s co-founder and CEO, told The Times of Israel: “Some 15 million people are treated for stroke every year and stroke is the main reason for disability, with 75% of patients suffering from long term disability.”
He added that data from a pilot study down by BrainQ showed 77% of the patients treated with BrainQ’s device “recovered to having little to no disability, which is a very striking result.” The study involved 25 stroke patients.
The 77% recovery rate impressed scientists worldwide. It was enough for the U.S. Food and Drug Administration to give the start-up company Breakthrough Device Designation. The Breakthrough Devices Program recognizes “certain medical devices and device-led combination products that provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.”
The program also gives BrainQ the chance to work closely with U.S. regulators to speed up the development process for the device, as well as final approval for its use. BrainQ hopes the device will also have benefits for those who have suffered a spinal cord injury or been diagnosed with Parkinson’s Disease or multiple sclerosis.
BrainQ conducted the study on patients who were in the 3-4 range on what is known as the Rankin Scale. The scale measures neurologic disability after a stroke. The levels are as follows.
- No symptoms
- No significant disabilities
- Moderate disability requiring some help
- Moderately severe disability, unable to walk or attend to bodily needs without assistance
- Severe disability – bedridden, requiring constant nursing care
- Death
The patients who participated in the pilot study used the BrainQ device for eight weeks. They saw their score on the Rankin scale drop from the 3-4 level down to a 0 or 1. Overall, 92% of study participants improved by two or more points on the scale. In addition, the patients experienced no adverse affects.
Dr. Jeffrey Saver, director of the UCLA Comprehensive Stroke and Vascular Neurology Program, told the Times that current interventions are effective only for a few hours post-stroke and apply to less than 10% of patients.
“This technology may have the ability to extend the time period in which corrective therapy can be delivered from hours to days and even weeks,” he said, “and be applicable to a much larger patient population.”
